Imaging technologies like Computed Tomography (CT), Magnetic Resonance Imaging (MRI), and ultrasound have become a standard part of medical care. These scans rely on patients being administered some kind of imaging molecule, known as a “contrast agent,” that will show up on the scans, allowing doctors to look inside patients’ bodies non-invasively to see what’s going on in the cardiovascular system or tumors.
UConn professor of pharmaceutics, Xiuling Lu, and her former Ph.D. student André O’Reilly Beringhs ’21 (PHARM), developed a novel nanocapsule in which Perfluorooctyl Bromide (PFOB) was encapsulated that makes several key improvements over current contrast agents on the market with promising applications for cancer diagnosis and treatment.
The nanocapsule uses a polymer created by Rajeswari Kasi, a UConn professor of chemistry. These droplets are like oil in water – they don’t readily combine with the surrounding solution. The nanocapsule uses the polymer to stabilize the nano droplets to suspend them so that they create the contrast X-Rays picks up on.
The team recently published their findings which showed that their nanocapsules could effectively image nanoparticle distribution in tumor models. This article was highlighted as the cover article for the journal Advanced Therapeutics.
There are many cancer therapies on the market, or in clinical trials right now that use nanoparticles. Often, cancer treatments have large variations in patient responses, and the same drugs that worked for one patient might not work for another.
Lu’s nanocapsules can help scientists see how well nanoparticle-based delivery systems for chemotherapy drugs would work in a particular patient. The nanocapsules will allow imaging techniques to determine the amount and distribution of the nanoparticles, signifying how the drug would accumulate in a heterogenous tumor.
“A lot of therapies, when they get to clinical trials, fail because the treatment is not simple, and the tumors are not homogenous, and all patients are not the same,” Lu says. “If we have some way to understand the tumors better, this can help direct a better treatment.”
This paper demonstrated that tumors could be successfully imaged using the nanocapsules. This imaging provided an outline of the tumor’s makeup and how nanoparticles accumulated in it. Further, the researchers demonstrated that the accumulation of the nanocapsules correlated directly with the accumulation of the chemotherapy drug PEGylated liposomal doxorubicin.
“With this imaging tool, if you gave a patient a dose of this imaging agent and observed that this patient’s tumor had limited contrast in the image, a similar nanomedicine would likely have limited accumulation there and it won’t be as effective,” Lu says.
Lu previously published her research findings in the American Association of Pharmaceutical Scientists’ journal AAPS PharmSciTech, which reported improved stability and a higher concentration than competitors. It also showed improved qualities when compared with Iohexol, the most commonly used contrast agent for X-Ray imaging in the clinic.
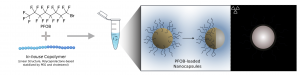
A graphic of the nanocapsule from Lu and Beringhs’ AAPS PharmSciTech paper.Another concern these nanocapsules address is that current market alternatives can only stay in the body for a very short time. For example, Iohexol only stays in the circulation for 30-45 minutes. This quick clearance rate is fine for studying the circulatory system, but is not ideal for imaging tumors. Lu’s nanocapsule, by contrast, has a nine-hour half-life. Because other molecules are very small, the body processes and expels them very quickly.
“Due to their nano size, they are larger than those small molecule contrast agents,” Beringhs says. “Because of that, they stay in the blood circulation longer, so they allow us to get very good imaging when it comes to the blood vasculature. These nanocapsules also extravasate into tumors and provide insights on the heterogeneous structure of solid cancers, which is valuable for treatment purposes.”
Lu’s invention would allow patients to be imaged multiple times without having to receive multiple doses of the contrast agent. This alleviates toxicity concerns and the effect other medications have on the distribution of the contrast agent, especially for older patients who undergo these processes more frequently.
While the researchers have only looked at CT scans, other studies have shown PFOB formulations are effective contrast agents for MRI and ultrasound. The researchers believe the nanocapsule would be more effective than current contrast agents in those modalities as well, given its higher concentration and stability.
“The value goes way beyond CT scans,” Beringhs says. “It can be used for a wide variety of imaging modalities.”
Another possible application of these nanocapsules is as an oxygen carrier. PFOB can dissolve a considerable amount of oxygen, making it useful as artificial blood replacements.
This feature is also relevant for cancer treatment. Tumor hypoxia, or lack of oxygen, causes resistance to chemotherapy, radiation, and immunotherapies. The nanocapsules could help deliver oxygen to alleviate this effect.
Lu is the co-inventor of four issued patents. Lu also founded Nami Therapeutics, a UConn Technology Incubation Program (TIP) startup that develops specifically designed nanoparticles for the targeted delivery of therapeutic agents.
UConn Technology Commercialization Services has filed patent protection for this technology. TCS is looking for collaborators/licensees in the industry to move this technology forward. For further information, please contact: techcomm@uconn.edu
Under the umbrella of UConn’s Office of the Vice President for Research, Technology Commercialization Services (TCS) works with innovators, entrepreneurs, investors, and industry partners to transform UConn discoveries into products, companies, and jobs that benefit society and fuel economic development. Through a coordinated approach between tech transfer, licensing, and startup teams, TCS provides service that enables success for faculty, business, Connecticut, and beyond.