Antibody treatments exist for many diseases, but require injections or IV infusions, which are painful, inconvenient and carry risks. Now, a team of researchers at the University of Connecticut has created a way to deliver multiple doses of antibodies over a period of weeks using a small, biodegradable skin patch instead.
“It solves a lot of problems,” says Thanh Duc Nguyen, an associate professor in the department of mechanical engineering and biomedical engineering, a joint department between the UConn Schools of Medicine, Dental Medicine, and Engineering.
Antibody therapies have been successful in treating HIV, autoimmune disorders such as multiple sclerosis, and certain types of cancer. But as Nguyen points out, administering antibody therapies can cause problems. Repeated injections can be painful, get infected, and scar veins. They also require a trained medical professional to administer, and the used needles are biohazardous waste.
The injections also need to be done frequently, often on a weekly basis, to maintain adequate levels of antibodies in the blood, as antibodies often have a short lifetime in the body. This can be a difficult schedule for patients to maintain if they lack easy access to transportation or live far away from the treatment center.
Now, Nguyen and co-authors Khanh Tran, a former UConn student now a post-doc at MIT, Eastern Virginia Medical School (EVMS) researcher Gustavo Doncel, and colleagues from EVMS and UConn, report a better way to deliver antibodies in the March 4 issue of Molecular Pharmaceutics.
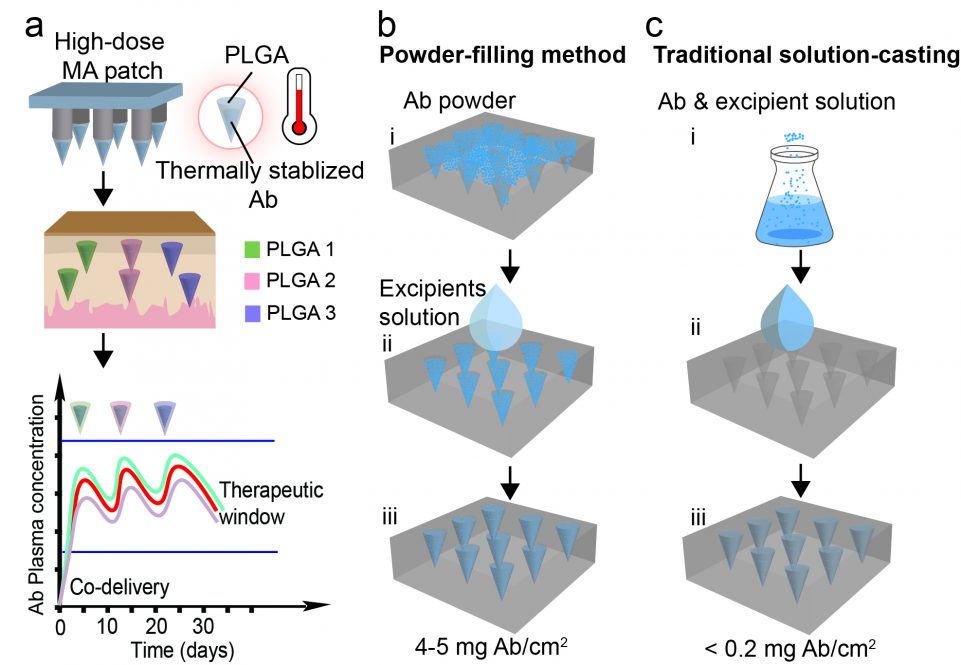
The team made a microneedle patch out of a biodegradable polymer. The patch has tiny needles that penetrate the skin only through the nerveless surface layer. Patches using the same basic idea are used to deliver some types of timed-release birth control and other medicines, but these patches won’t work for antibodies made of proteins that easily decompose if they get too dry or too warm. Nguyen, Tran, and their colleagues had already figured out how to stabilize proteins using sugars instead of water. This time, they also had to figure out how to deliver large doses of antibodies multiple times over a period of weeks.
They developed a patch that uses a biodegradable polymer as a shell, filled with a stabilized powder of antibody. The polymer biodegrades at a known, controllable rate (tailored biodegradable polymers are a speciality of Nguyen’s lab.) As it degrades, it slowly delivers the antibody into the patient’s bloodstream. Additional layers of polymer that take longer to biodegrade can deliver additional doses of antibody over a period of weeks.
The team tested the patch in rats, and found it could maintain fairly steady levels of therapeutic antibodies in the rat’s bloodstream for over a month.
The next step is to test the patch in a larger animal, and then in humans. If it works, it would make it much easier and safer to get antibody treatments. A self-administered patch would cut down on visits to a clinic, reduce the risk of infections from needle sticks, and eliminate the biohazardous waste from injections.
The team at EVMS already has a large animal program studying HIV, and they hope to test the biodegradable patches there. If all goes well, Nguyen has a startup, SingleTimeMicroneedles (STM), that is already working to commercialize single-use, biodegradable microneedle patches.
This research was funded by The U.S. President’s Emergency Plan for AIDS Relief, and the Institute of Materials Science at UConn.