Damaged nerves of the brain, eye, and spinal cord cannot grow back. But specific gene therapies might be able to change this, leading to treatments for paralysis and other forms of nerve damage, UConn Health researchers report in the October issue of Experimental Neurology.
Axons are the long arms of nerve cells that reach from our extremities to our spinal cord, and from our eye to our brain. Injuries that smash or sever axons—and often the large bundles of axons that we commonly call nerves—can cause paralysis, blindness, lack of sexual function, or other devastating outcomes. Most of the time, these central nervous system axons don’t repair themselves, and we have no good treatments for this.
Axons fail to regenerate for several reasons. Some of them have to do with the environment the axon grows in, but another reason is that the ability to grow is lost as the nervous system matures during and after birth. The loss of key proteins prevents regrowth once an organism matures, reports a team of researchers at UConn School of Medicine.
“We are deciphering the mechanism of axon regeneration piece by piece,” says School of Medicine neuroscientist Ephraim Trakhtenberg.
Graduate students Jian Xing and William Theune, postdoctoral research fellow Agnieszka Lukomska, and colleagues in Trakhtenberg’s lab found that a group of genes that are generally dormant in mature nerve cells are key to axon growth and regeneration. The team looked at a specific group of 80 genes, called ribosomal protein genes, and tracked how they changed during development. The researchers found that all 80 ribosomal genes progressively quieted, becoming less active as nerve cells matured. Ribosomal protein genes are needed for making ribosomes, which are tiny factories in the cell responsible for making proteins. Protein production is key to all forms of growth, whether it’s muscle growth after hitting the gym, or the development of a tiny embryo into a fully grown individual. As embryonic nerve cells finish growing and mature, the demand for these ribosomes diminishes, and as a result the ribosomal genes become nearly inactive.
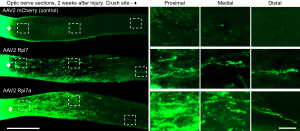
The researchers decided to look at two members of this gene family, Rpl7 and Rpl7a, which had previously been found to be associated with neurodegenerative diseases. The researchers altered nerve cells so that instead of these genes quiet, they would be robustly expressed in the adult cells. The researchers then crushed bundles of axons from these cells, modeling damage to the optic nerve (the nerve that connects the eye to the brain). Damaged axons of the optic nerves never grow back in adults, causing blindness. But the neurons that had been altered to keep producing Rpl7 and Rpl7a began growing axons again after injury.
“By leveraging our understanding of embryonic development, we can reinstate the genes we find to be necessary for axon growth in adults after injury,” and “by studying how genes change across development, we can identify promising candidates for treatment of nerve injury and disease,” say graduate students William Theune and Jian Xing and postdoctoral research fellow Agnieszka Lukomska.
The results suggest that the 80 ribosomal protein genes that are found at low levels in adult nerve cells may be very important to axon regeneration. The Trakhtenberg lab has previously identified other factors in the nerve environment that prevent nerve regrowth, and this paper adds another layer of understanding at the level of molecular mechanisms.
“Now we need to look deeper at what is happening inside neurons that can accelerate axonal growth and overcome the environment more efficiently,” Trakhtenberg says.
This work was supported by grants from the UConn School of Medicine Start-Up Funds, and the National Institutes of Health National Eye Institute.



