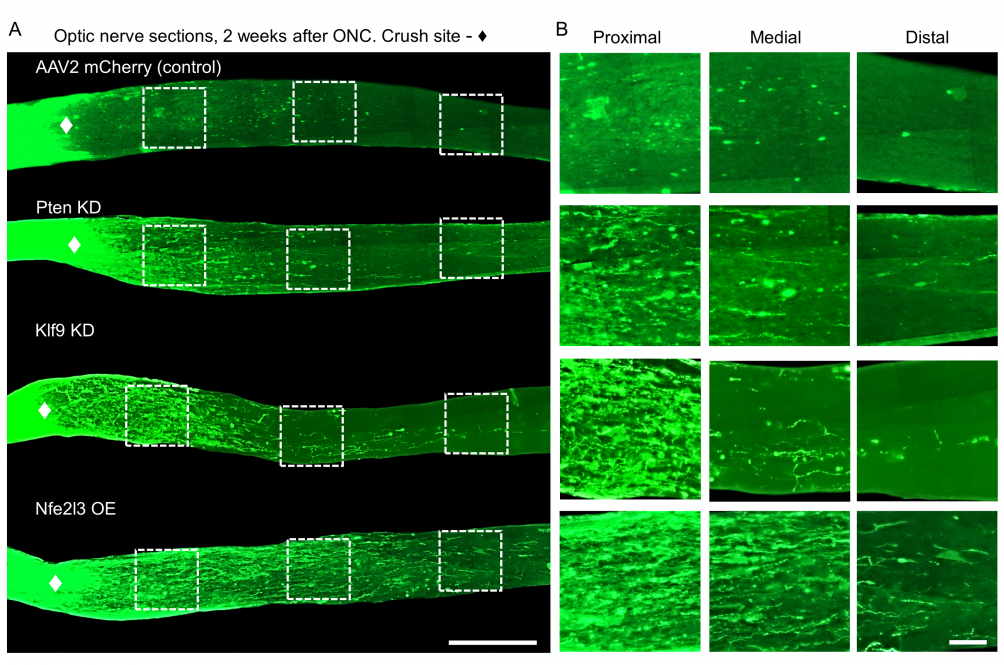
Damage to the optic nerve can lead to irreversible blindness. A newly investigated regeneration factor could change that, UConn researchers report in the May 2024 issue of Experimental Neurology.
Blindness and vision impairment due to optic nerve damage affect more than 3 million people in the US alone, according to the Centers for Disease Control (CDC). The most common reason for that damage is glaucoma, a family of eye diseases that affect the flow of liquid in the eye, eventually damaging the long bundle of cells that connect the retina to the brain. That bundle of cells is the optic nerve. They don’t grow back after being damaged, leading to permanent vision loss.
Now, a team of researchers in the lab of UConn School of Medicine neuroscientist Ephraim Trakhtenberg have shown that a protein previously thought unimportant can stimulate regrowth of nerve cells. The protein is called nuclear factor erythroid 3 (Nfe3), and it is unique to nerves originating in the retina. Normally it is not produced by adult neurons.
Trakhtenberg and colleagues suspected that Nfe3 played some specific role in nerve growth, and that they might be able to harness it to regrow nerves after injury. They took adult mice with crushed optic nerves and stimulated Nfe3 production using a gene therapy. Just as the researchers hoped, individual nerve fibers in the mice’s damaged optic nerves began to regrow (see image.)
The regrowth was significant, about as good as the best regenerative factors previously known. But Nfe3 is different, and more promising, than some of those other regeneration factors. Some of these other regenerative factors have downsides: they tend to cause inflammation, or risk inducing tumors. Nfe3 has no similar problems.
“This opens a whole new novel realm of research. It could help glaucoma and other types of nerve damage,” Trakhtenberg says.
The next step is to observe the regenerating nerve cells for several months to see if they eventually reconnect to the brain, a necessary step to restore sight. If they do reconnect to the brain, Nfe3 could truly be a potential treatment for cases of optic nerve damage. It could also be potentially prescribed as a preventative, to stop visual loss in diseases that slowly damage the retinal neurons and the optic nerve over time. If the factor is successful in repairing the optic nerve, it might also be useful in treating paralysis and other forms of nerve damage in the brain and spinal cord.
This work was supported by a grant from the National Institutes of Health National Eye Institute.