Severe bacterial infections can push the human body into sepsis, a life-threatening cascade of inflammation and cell death that can be difficult to cure. Now, researchers at UConn Health have discovered how bacteria trigger such a dangerous inflammatory reaction from our body.
Bacteria are not supposed to be in the bloodstream. If they get into the blood, the body mounts a vigorous response. And if bacteria start invading not just the blood stream, but cells in the blood stream, the body’s response is more than vigorous – you could call it apocalyptic. A cell that detects that it’s been invaded by bacteria destroys itself, bursting open and spewing bits of the invading bacteria, along with inflammatory chemicals that signal to every immune cell within shouting distance “Problem! Emergency! Get over here and check this out!”
“It signals to the rest of the body that there is an overwhelming infection. It’s very serious,” says Vijay Rathinam, an immunologist at UConn Health. It sometimes leads to a runaway series of cell suicides that cause sepsis, with inflammation, fever, and low blood pressure, which can be deadly.
Scientists and doctors have long puzzled over this; why does the body mount such a dangerous, self-damaging response? In the May 19 issue of the journal Cell, published online today, Rathinam and colleagues at UConn Health’s Department of Immunology propose an explanation: The cells aren’t really being invaded. They just think they are.
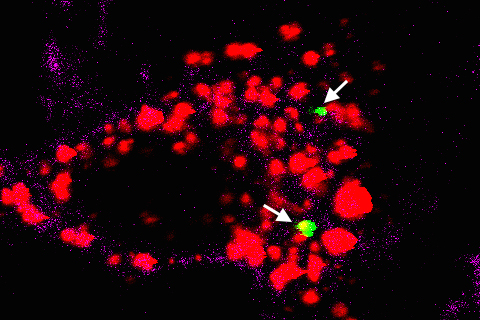
What really happens is this: yes, there are a certain class of bacteria, called gram-negative bacteria, in the bloodstream. Gram-negative bacteria secrete little vesicles. The vesicles can interact with human cells, gaining entrance into the cell. Once they get inside, they spill their payload of lipopolysaccharides (LPS). LPSs are potent molecules that set off alarm bells whenever they are encountered by the immune system. When the cell realizes it has LPSs inside of itself, it thinks a bacterium has slipped past its defenses, and it commits suicide, spilling out its inflammatory cytokines and alerting the immune system.
This all makes sense, except when you think about it from the bacteria’s point of view. The bacteria don’t want to kill their host; they perhaps want to survive and multiply. So why do they send out little mail bombs that destroy the host’s cells?
Sivapriya Kailasan Vanaja, an assistant professor in the Department of Immunology and lead author of the paper, says the purpose of the vesicles isn’t really to kill the cells – that may be just a side effect. Vesicles are the tools that cells, including human cells, use to communicate with each other. Bacteria use them the same way, to communicate with and influence the functions of neighboring bacterial and human cells. For example, vesicles secreted by bacteria can modify nearby cells in such a way as to favor bacterial replication. Vesicles can also do other tasks. For example, vesicles act as a sponge to absorb molecules toxic to bacteria, such as antibiotics.
“Bacteria ramp up production [of LPS-filled vesicles] when they are in a hostile environment,” says Rathinam. “In a way, the death of the host cells is just collateral damage.” But it often creates a vicious cycle for sepsis sufferers. The more hostile the microenvironment becomes, the more vesicles the bacteria produce. The more vesicles the bacteria produce, the more cell suicides, and so the more inflammation and fever.
But the vesicles could also turn out to be a tool for researchers and physicians. It might be possible to inhibit the vesicles’ interactions with the body, for example, stopping the runaway septic reaction. Or as part of a vaccine, the vesicles might be good. The right quantity of LPS-filled vesicles could trigger a little inflammation, attracting immune cells and enhancing the vaccine’s efficacy. On the other hand, the LPS detection inside the cells might be dangerous, increasing the vaccine’s toxicity. In this case, researchers might devise a strategy to avoid it.
The research “opens up a whole new avenue to understanding this,” says Kate Fitzgerald, professor of medicine and director of the Program on Innate Immunity at the University of Massachusetts medical school. “Only gram-negative bacteria can activate this pathway – it certainly sheds light on one way sepsis can happen.” Fitzgerald says Rathinam’s work could lead to new, targeted therapies.
The next step for Rathinam’s group is to figure out what it is about vesicles that allows them to infiltrate cells and gain intracellular access so easily. Once they do that, they can figure out if there are any host molecules that enable the process, and that will give clues to therapeutic treatments, Rathinam says.
UConn Health immunology graduate students Ashley Russo, Bharat Behl, and Ishita Banerjee contributed to this work, and it was supported in part by a grant from the National Institutes of Health, as well as by a grant from the Charles Hood Foundation.